
Deep Cycle Batteries
Deep Cycle Solar Batteries for your Solar Power Systems
![]() Photovoltaic solar panels produce electricity whenever the sun shines on them. Stand alone (off-grid) and battery backup grid connected PV systems require a single battery or a group of Deep Cycle Batteries called a “battery bank” wired together to store the generated solar energy.
Photovoltaic solar panels produce electricity whenever the sun shines on them. Stand alone (off-grid) and battery backup grid connected PV systems require a single battery or a group of Deep Cycle Batteries called a “battery bank” wired together to store the generated solar energy.
The photovoltaics charge these deep cycle batteries during the daylight hours so that you can have electricity at night or on cloudy days. Then storage batteries are an essential part of any stand alone or grid-tied alternative energy installation and which themselves can determine the DC operating voltage of the whole photovoltaic solar system.
Deep Cycle Batteries consist of a collection of individual smaller 2-volt cells which store the electrical energy produced by the PV panels that are not immediately consumed by the load. Battery cells allow this electrical energy to be converted into chemical energy, stored within the cell, which is then converted back into electrical energy as needed.
A battery consists of one or more of these cells, electrically connected together in series or parallel, or both, depending on the required output voltage and amperage. Then we can define a battery for use in a solar power system as a collection of cells which store electrical energy in the form of chemical reactions.

In the world of batteries, Deep Cycle Batteries are classed as “secondary batteries” because they can be continuously charged-up and discharged producing what is know as the batteries charging cycle.
Deep cycle batteries are called secondary batteries because the chemical reaction that produces and stores the electrical energy on its lead plates is fully reversible, as opposed to standard “primary batteries” that can only be used once and then thrown away once they are fully discharged.
In other words, primary batteries such as those small alkaline power batteries used in remote controls or cameras can only be discharged or used once and are then discarded (thrown away) polluting the environment, while secondary batteries called “rechargeable’s” such as NiCad or Lithium Ion batteries used in laptops etc, can be discharged and recharged many times over and are therefore much kinder to the environment.
In this tutorial about “deep cycle batteries”, we are mainly interested in true deep cycle batteries which are ideal storage devices for solar energy systems and can be characterised (in addition to their ability to be recharged) by high power density, high discharge rate, flat discharge curves, and good low-temperature performance.
But not all secondary batteries are the same. There are many different types of batteries available, with each type designed for a specific applications. Although similar to ordinary car batteries, deep cycle batteries used in solar power systems are especially designed for the daily charging and discharging cycles they need to endure. The most common type of secondary batteries used in solar PV applications are typically maintenance free “lead acid batteries”. Since this type of rechargeable battery is the most cost effective and readily available types for use in a home energy storage system.
Lead acid batteries are one of the oldest and most common battery technologies and are made up of three basic parts: a negative electrode, a positive electrode, and an electrolyte. The black coloured negative electrode is made of lead (Pb), and the red coloured positive electrode is made of lead dioxide (PbO2). An insulator or separator is used to electrically isolate the two electrodes. The electrolyte used to cause the chemical reaction between the two electrodes is dilute sulfuric acid (H2SO4), which is an acidic liquid that provides the sulfate ions for the discharge reactions.
Wet lead acid batteries are usually the least expensive, but require adding distilled water (H2O) occasionally to replenish water lost in evaporation during the normal charging process. Then basically, the negative electrode gives up electrons to an external load, and the positive electrode accepts electrons from the load and the electrolyte which is typically a liquid in deep cycle solar batteries provides the pathway for charge to transfer between the two electrodes as shown.
Deep Cycle Solar Battery Cell
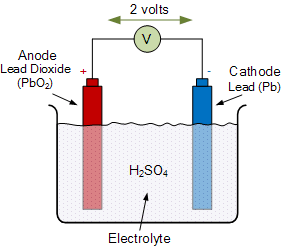
A basic functional diagram of a typical lead acid cell is shown on the right. A plate of solid lead serves as the negative electrode, and a plate of lead dioxide serves as the positive electrode. Both electrodes are immersed in a sulfuric acid solution called the “electrolyte”.
The result is that a potential difference, more often called a voltage, develops between the electrodes and it is this voltage that is used to drive current through an external load.
The maximum deliverable current generated by the lead-acid battery depends on the mass and volume of the cell. In a lead acid battery made from lead acid cells connected in series (negative-to-positive), the overall terminal voltage depends on the number of cells connected together within the battery.
Parameters associated with deep cycle lead acid batteries are:
- Voltage – Voltage is electrical pressure. A standard car battery is 12 volts. This voltage is the addition of the six (6) smaller lead acid cells connected together in series which make up a larger 12V battery. Each individual lead acid cell has a voltage of about 2 volts. Battery banks used for alternative energy systems are usually connected together in series to produce DC voltages of 12, 24, 36, or 48 volts.
- Current – Current is the flow of electrons. The rate of this flow per unit time is called an ampere. Electrical current comes in two forms: direct current (DC) and alternating current (AC). Batteries store power as direct current (DC) which is used for lighting or to power the inverter which changes it into alternating current. The maximum deliverable current from deep cycle batteries is the highest current a battery can drive through a load without its terminal voltage dropping significantly due to the batteries internal resistance, and without causing the battery to overheat. Deep cycle batteries are connected together in parallel to increase the available output current.
- Capacity – Battery capacity is the amount of energy a battery contains and is usually rated in Ampere-hours (Ah) at a given voltage. So a battery with a rating of 1,000 ampere-hours can deliver 100 amperes for 10 hours, or 10 amperes for 100 hours, or 1 ampere for 1000 hours and so on. To determine the total amount of power a deep cycle battery can deliver, multiply the ampere-hours (Ah) by the terminal voltage. The storage capacity of an average car battery is about 40 to 85 ampere-hours.
Deep cycle solar batteries used for alternative energy installations have from about 200 to 2,500 ampere-hours capacity. The specified capacity of a battery is determined by how much energy is needed and for how long must the battery supply this energy. The more electrical energy that is consume the larger the battery must be and the solar power source must therefore be larger to recharge the larger battery.
A batteries state of charge is a percentage value that indicates the amount of electrical energy remaining in the battery. For example, a 1,000 ampere-hour battery at a 90% state of charge will contain 900 ampere-hours of energy. At 50% state of charge it will contain 500 ampere-hours of energy and so on. A battery which has only 20% or less state of charge is considered to be completely discharged or is possibly faulty.
True Deep Cycle Batteries for Solar Applications
Lead acid batteries are the only batteries suitable for alternative energy systems but the continuous cycle of discharging a battery into a load (night time hours) and then recharging the battery (sunlight hours) many times over requires a different type of battery as not all lead acid batteries are the same. The most important requirement of a battery is whether it is a Deep Cycle Battery or a Shallow Cycle Battery.
Consider automotive starting batteries. Automotive vehicle batteries are cheap to buy but are designed to provide high amperes of current for a very short period of time (less than 10 seconds) to operate the starter motor and turn over the engine. These are commonly called “cranking amps”.
After the car has started, the battery is then trickle charged by the cars alternator. Even on cold frosty mornings the cars battery will only discharge down to about 90% of its rated full capacity at start-up. Thus automotive batteries are designed for very shallow cycles between 100% to 90% state of charge.
As a car battery is designed to deliver high currents for very short periods of time it is therefore made of many thin lead plates giving a large surface area for the chemical reaction to occur.
These thin lead plates do not have the necessary mechanical strength for repeated cycling over a period of many years and wear out very quickly after only 200 to 400 cycles. Therefore shallow cycle car batteries which although they work, are not designed for a long term solar power system which requires a much deep cycling service.
Deep Cycle Batteries on the other hand are designed to be repeatedly charged and discharged by as much as 80% of their full capacity (100% to 20% state of charge) without sustaining any serious damage to the cells before recharging, making them an ideal choice for solar photovoltaic system as well as marine applications, golf buggies, fork lift trucks and other such electric vehicles. Although deep cycle batteries uses the same chemical reactions to store energy as their automotive battery cousins, deep cycle batteries are made very differently.
The physical size of a deep cycle battery is much larger than a regular car battery due to the construction and size of the lead plates (electrodes). These plates are made of solid lead usually doped with Antimony (Sb) and are many times thicker than the thinner sponge type plates of a car battery. This means then that deep cycle batteries can be repeatedly discharged almost all the way down to a very low charge and it is not uncommon for deep cycle batteries to be emptied (discharged) to as much as 20% of their total capacity before energy ceases flowing from the battery.
Deep cycle batteries are designed specifically for storing the energy generated by a photovoltaic PV systems and then discharging this stored energy for use on a consistent, daily basis. One of the main requirements for deep-cycling batteries for solar applications is maximum cycle life, that is how many times can the battery be charged and the discharged or deep cycled.
Deep cycle batteries used in alternative energy applications should last for more than five years and in many cases last well over ten years but it must be cycled properly. Even though these batteries are designed to withstand deep cycling, deep cycle batteries will have a longer life span if the cycles are shallower, for example, 100% to 50% state of charge as opposed to a 100% to 20% state of charge.
Deep Cycle Battery Charging
Deep cycle batteries can be recharged in a variety of different ways. The most common method is the use of an external charging unit connected to a source of electrical power such as a wall socket. In an alternative energy system, the batteries are charged by solar panels via a battery charge controller that ensures the maximum output from the solar panels or array is directed to charge the batteries without overcharging them.
A deep cycle lead acid battery can generally be charged at any rate that does not produce excessive gassing, overcharging, or high temperatures. The battery absorbs very high current during the early part of the charge when its state of charge is at its lowest, but there is a limit to the safe current as the battery becomes fully charged.
12 Volt Battery Charge Condition
| Open Circuit Voltage | State of Charge |
|---|---|
| 12.65V | 100% |
| 12.58V | 90% |
| 12.55V | 80% |
| 12.48V | 70% |
| 12.40V | 60% |
| 12.32V | 50% |
| 12.24V | 40% |
| 12.10V | 30% |
| 11.90V | 20% |
| 11.70V | 10% |
| 11.30V | 0% |
If a deep cycle lead acid battery is connected to a load for a long time without maintaining its charge, the battery gradually discharges its power into the load.
As the current gradually decreases, and the electrodes become coated with contaminants as the specific gravity of the electrolyte changes.
This contamination inside a lead acid battery is called “Sulfation”. Eventually, all the chemical energy contained within the electrolyte is converted into electrical energy.
At some point the battery process stops as the battery can no longer deliver power to a load. The current drops to zero, and a potential difference no longer exists between the electrodes in the cells.
The amount of power that a lead-acid cell battery can deliver is a function of the electrolyte (specific gravity and purity) and the quality of the negative lead electrode and positive lead-dioxide electrode.
Over time, sulfation interferes with the ability of the battery to accept, store and deliver a charge, and if left unchecked the quality of the battery components degrade, and the battery performance decreases rendering the battery useless far short of its designed life. The fastest way to destroy any lead acid battery is to allow it to discharge completely and let it stand uncharged for a long period of time. So due to chemical interactions inside a lead battery, it must be used on a regular basis or sulfation of the plates will occur.
However, if a current is driven through the battery by connecting an external power source (this is commonly called recharging, and is simply the inverse of when the battery delivers its power to a load and in this case, the battery is now the load), for a period of time (negative-to-negative and positive-to-positive), the electrochemical energy in the battery is restored, and the battery can be used again.
The cycle of discharging a battery into a load and then recharging the battery can be repeated many times over the useful life of a battery but use a hydrometer regularly to check the specific gravity and top-up with distilled water when fully charged.
Connecting Deep Cycle Batteries
We now know that the basic building block of a lead acid battery is the 2-volt cell. A battery bank used in off-grid and stand alone solar power, or wind power system is a collection of connected 2 volt cells, 6 volt batteries, or 12 volt batteries that supply power to the household in case of mains power failure or low production from renewable energy sources or a solar array. Individual deep cycle batteries are wired together in series to produce 12 volt, 24 volt, or 48 volt configurations.
Deep cycle batteries can also be connected together in parallel to increase the current capacity of a battery bank. The battery bank supplies DC power to an inverter, which produces AC power that can be used to run appliances. The decision to select either a 12 volt, 24 volt, 36 volt or 48 volt battery bank will be determined by the inverters input, the type of battery you select, and the amount of energy storage you require.
Below shows an example of connecting batteries together of different voltages, such as 6 volt and 12 volt batteries, to produce a 24 volt battery bank. Any number of batteries can be connected together in series to produce an output voltage that is a multiple of the battery voltage. In our example this is 2 x 12 volts = 24 volts. Likewise, batteries connected together in parallel increases the current by the number of branches. However, it is better to limit the number of connect branches to a maximum of three (3) as parallel battery banks tend to circulate unwanted currents from branch to branch.
Deep Cycle Battery Wiring
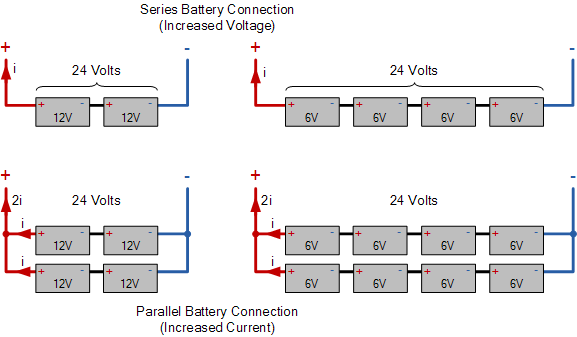
Maintenance of Deep Cycle Batteries
One final comment about battery maintenance. Lead acid deep cycle batteries are the most dangerous part of any solar or wind power system. Gloves, eye protection such as goggles and masks as well as old clothes must be worn when handling lead acid batteries and electrolyte as “battery acid” both burns and irritates skin and eyes.
Also hydrogen gasses and fumes emitted during charging of these lead acid deep cycle batteries is both an irritant and potentially explosive so ventilate the battery banks and area well at all times. Clean any spillages of electrolyte on or around the batteries and check the battery terminals and cables for tightness lubricating with petroleum jelly if needed. With proper care and maintenance, deep cycle lead acid batteries will have a long service life in any solar powered PV system.




![ExpertPower 12v 33ah Rechargeable Deep Cycle Battery [EXP1233 ]](https://m.media-amazon.com/images/I/31eYTwHWf2L.jpg)





These batteries are designed to store and deliver energy efficiently, ensuring a steady power supply even during cloudy days.
Thanks for sharing the valuable and informative blog.
I have a single 2 volts deep cycle battery. Is it possible to connect it to 12 volt solar panels and inverter for use as family power?
Basically no, a single 2 volt battery cell would not be enough for family power. Obtain another five battery cells of the same type and amperage and connect them together in series. Then you will have a 12 volt system for charging and power.
Very helpful for explaining the basics of the 3 different types – how much is the cost difference between a Lead Acid, AGM and Lithium battery of the same capacity be though?
I highly recommend this company to everyone! From start to finish, the entire company was extremely helpful and answered all my questions. They took care of everything from dealing with my township, getting everything set up with my utility company and even set me up with the state credits. This solar photovoltaic system was installed in about a month from signing and my wife and I love the way our system looks on the front of our house.
Thanks for this valuable information…
My Panels are 24v, 300w that go to 24v charge controller. I am thinking of using the in series, 4 – 6V batteries which will in turn be wire to my 3000w, 24v inverter. First, is this feasible and second where in the series do the charge controller and inverter connect to the 4 – 6v batteries?
Four 6V batteries connected in series would give you a 24 volt system, then your 24V panels and controller would connect across all batteries.
I am new at this. I am installing 100W solar system with 12V battery. Plan on powering lights at night. I will be installing in line fusing between solar panel and controller (8amp)and between battery and controller (30 amp). How do you feel about inline switches on these circuits as well?
Solar switches and isolators add safety to a solar system by allowing parts of the system to be disconnected from possible high voltages. It is advisable to install switches which are specifically designed for DC or solar use and IP66 rated.
While flooded lead acid batteries have been the go to for years, there are a number of deep cycle technologies available. AGM and LiFePo are two that come to mind. There are pros and cons for any battery system but limiting the article to only flooded lead acid does not reflect the current state of what’s available.
Energy storage systems are constantly developing and changing as new technologies become available, so any article about a particular technology, would in time, become out of date, but still relevant. This tutorial is about deep cycle flooded lead acid batteries and its content reflects that.
I’m doing a project myself to power up some devices with a total of 155 Watts by using 150 Watts solar panel. If I want to use the devices during night time hours, I will have to connect them with a battery. With my current setup (150W solar panel), do you think it is able to charge the battery? Or I need higher wattage solar panel to make everything works (recharging and power my devices at the same time)
Thank you.
Well that depends on the Amp-hour rating of your battery/batteries and the amount of electrical power drained from it/them during the night.
I have 2 Panels. Rated 62.5 Watts – located on the South ( morning sun) and South ( evening) directions. I measure on average 65 Victor from my array….and my 100 ah Battery is never fully, charged ( max 12.2).
Mppt 1010 (10amp)
I still have more of same panels and Battery.
What do you advise.
More Panels?
If your existing configuration is not capable of fully charging your battery during the day, then yes you may need more panels.
Thanks for sharing your thoughts on deep cycle batteries.
Regards