
Photovoltaics Turn Photons into Electrons
PV Cells Turn Photons into Electrons
![]() Photovoltaic cells, or PV’s for short, are magical things which convert light energy, usually from the sun into electrical energy through a process called the photovoltaic effect. This photovoltaic effect occurs in materials known commonly as semiconductors, and solar cells are made from semiconducting materials such as silicon (Si), the second most abundant material in the world after oxygen, which is used to convert the sunlight into electricity. That is photovoltaics turn photons into electrons.
Photovoltaic cells, or PV’s for short, are magical things which convert light energy, usually from the sun into electrical energy through a process called the photovoltaic effect. This photovoltaic effect occurs in materials known commonly as semiconductors, and solar cells are made from semiconducting materials such as silicon (Si), the second most abundant material in the world after oxygen, which is used to convert the sunlight into electricity. That is photovoltaics turn photons into electrons.
This conversion process as effective as it is, produces no harmful waste or emissions to the environment making it a clean, green and effective for of energy generation. Then a photovoltaic cell, also known as “solar cell”, is a semiconductor device that generates DC electricity when light falls on it.
Photovoltaics turn photons into electrons by what is called the “photovoltaic” effect, (photo meaning light, and voltaic meaning electricity). In the photovoltaic effect, rays of photons in the sunlight strike the surface of the silicon semiconductor material liberating free electrons from the materials atoms.
There are a variety of different materials that a solar cell can be made from with silicon being the most common. Whatever the material, all photovoltaics convert solar radiation into electrical direct current (DC) power, which can then be used to power electrical devices.

The photovoltaic solar cell is generally made from modified silicon crystals such as crystalline and amorphous silicon, or other semi-conductive materials, that absorb and convert sunlight into electricity.
Certain doping chemicals are added to the silicon composition to help establish a path for the freed electrons to flow through. This creates an electron flow called a DC current.
Semiconductors are non-metallic materials, such as germanium and silicon, whose solar cell I-V characteristic lie somewhere between those of a conductor, which offer very little resistance to the flow of electrical current, and an insulator, which blocks the flow of electrical current almost completely. Hence the term “semiconductor”.
The silicon based material is called a semiconductor, because the device conducts electrons in one direction only from negative to positive.
Crystalline silicon has been the main material used to manufacture solar photovoltaic cells of the last decades with polycrystalline silicon and amorphous silicon leading the way. The recent increase in interest in home solar energy and photovoltaic cells has advanced solar thin-film technology greatly with cadmium telluride and copper indium sulphide now being used in the home renewable energy applications.
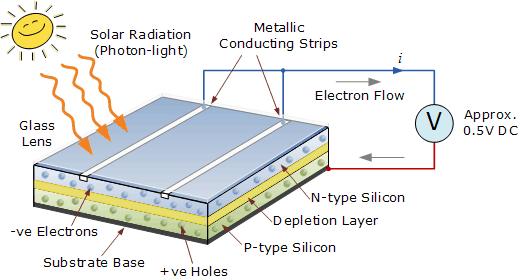
Photovoltaic cells consist, in essence, of a junction between two thin layers of dissimilar semiconducting materials. One layer of silicon is treated with a substance to create an excess of electrons. This becomes the negative or “N” type semiconductor layer. The other layer is treated to create a deficiency of electrons, and becomes the positive or “P” type semiconductor layer.
N-type semiconductors are made from crystalline silicon that has been “doped” with tiny quantities of an impurity atom (usually phosphorus) in such a way that the doped material possesses a surplus of free electrons, hence the term negative-type semiconductor. P-type semiconductors are also made from crystalline silicon, but are doped with very small amounts of a different impurity atom (usually boron) which causes the material to have a deficit of free electrons.
These “missing” electrons in the semiconductor lattice are affectionately called “holes” and since the absence of a negatively charged electron can be considered equivalent to a positively charged particle, silicon doped in this way is known as a positive-type semiconductor.
When these dissimilar semiconductor materials are assembled together with conductors, the arrangement becomes a light-sensitive PN junction semiconductor that sets up an electric field in the region of the junction and which we know commonly as a photovoltaic solar cell. Thus when similar such solar cells or PV’s are combined into solar panels, it results in a much higher electricity generating capacity.
Photovoltaic Solar Cell Construction
So How do Photovoltaics turn Photons into Electrons
Sunlight acting as a fuel carries energy into the photovoltaic cell. When a photon particle from the sunlight strikes the surface of the silicon solar cell or the doped structures made up of silicon-phosphorus or silicon-boron, the photons of the absorbed sunlight knocks loose and dislodges electrons from the silicon atoms of the cell transferring the energy and exciting them.
This excitation of the electrons causes them to become released from its parent atom and moves to a higher valance level. As there are billions of photons striking the cell every second, there are lots of electrons knocked loose.
Eventually, the excited electron gets kicked out of the atom allowing it to roam freely around the semiconductor material. As one side of the PN junction has a “lack of electrons” (holes) while the other side of the junction has an “excess of electrons” these free electrons move through the junction, creating and filling in holes in the cell.
It is this movement of electrons and holes that generates electricity and as long as there is light striking the cell, there will be electrons flowing out of the cell. The physical process in which a PV cell converts sunlight into electricity is known as the photovoltaic effect.
The electrons freed by the interaction of the sunlight with the semiconductor material creates an electron flow as the free electrons move together around an external circuit. The generation of electrical power requires both voltage and current. So in order to produce power, the PV cell must generate voltage as well as the current provided by the flow of electrons.
The photovoltaics cell voltage is provided by the internal electric field set up by the PN junction. This photovoltaic cell junction can be thought of as a small battery producing a fixed output voltage of about 0.5 to 0.6 volts.
A single silicon PV cell typically produces an electric current of about 3 amperes, with one single photovoltaic solar cell producing up to 1.5 watts of power. Depending on the semiconductor materials used, some PV cells can produce more power than this, and some less. Thus 36 cells connected together in series have enough voltage and power to charge 12 volt batteries or run pumps and motors.
Photovoltaics Turn Photons into Electrons Summary
Sunlight is clean, easy to harness and is readily available worldwide, and as its from the sun, it will continue to be freely available for many millions of years to come. When photons from sunlight strike a solar cell, they transfer their energy to the semiconductor material, freeing electrons and the generation of electricity. This process is the basis for how photovoltaics turn photons into electrons, providing a renewable and sustainable source of electricity.
Solar power is clean, green and effective and with today’s efficient solar panels or photovoltaic cells easily available in the marketplace, home owners can have free and clean energy for many years to come with the only cost involved being in the cost of a solar panel itself, making solar power an ideal choice as part of a home based renewable energy system.
Photovoltaic cells convert one kind of energy (sunlight) into another (flowing electrons). Converting sunlight into electricity is also very environmentally friendly, as it produces no pollution or waste by-products, no air or water pollution, and is completely silent making it ideal for a greener future.
Photovoltaic cells are very modular so you can install as small or as large a PV system as you need. The simplest photovoltaic cells power watches and calculators, while larger more complex solar panels and arrays can be used to light houses and provide power to the electrical grid.
To learn more about how Solar Cells Turn Photons into Electrons and how you can use photovoltaic solar panels to generate free electricity for your home, or to obtain more information about the various solar panel designs and systems available, or to just explore the advantages and disadvantages of using solar panels and arrays, then Click Here to order your solar guide from Amazon today about see how solar panels and arrays can be used in your home or caravan to generate free, clean and green solar electricity using the power of the sun.











What chemical effect happened in depletion layer after photons strike on it in PV cells that converts photons into electricity
Electrons are released
All the electrons on an atom can’t be knocked loose without eventually running out. If the solar cell continuously knocks off electrons to harness as a current, how do the material atoms not run out of electrons after some time? How does the cell replace the electrons in the atoms?
Firstly there are billions and billions of free electrons due to the doping of silicon as a semiconductor material. As one electron gets knocked out of its orbit by a photon, the gap is immediately replaced by another. The flow of these loose or free electrons around a circuit being pushed forward by a voltage is called an electrical current.
I don’t get it either. If there are fewer electrons in a solar cell at the end of the day how long can it last?
Dave
Photovoltaic solar panels are made to last more than 25 years
Such explanation depends on a silly definition of electric current as flow of electrons. The electric current represents a flow of energy; i.e. waves. Tesla succeeded in transferring electric power in space. Accordingly; it is flow of waves.
Do you get my point. Look at one of my articles:
S. Abdelhady, “Comments on Einstein’s Explanation of Electrons, Photons, and the Photo-Electric Effect,”Applied Physics Research, 3 (2), pp. 230-240.
Thanks! This will be very useful for my physics assignment 🙂
Can those atoms of Silicon whose electrons have been freed, regenerate or renew themselves (to their original state) when the solar radiation impinging on them ceases? That is to say, is the creation of a electron-hole pair due to incident photon a non-reversible process inflicted on some atoms of Silicon?
Thank you so much! I have been looking for an article like this for my project! Awesome job, keep it up!